Highlight
Development of a Probe for Membrane Cholesterol Accessibility
Achievement/Results
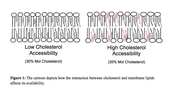
At the University of Massachusetts Amherst, Benjamin Johnson (ICE IGERT trainee) and Alejandro Heuck (ICE IGERT Advisor) have created a probe for membrane cholesterol accessibility based on the cholesterol-binding properties of a pore forming toxin called Perfringolisin O (PFO). Cholesterol is a primary constituent of mammalian cell membranes, helps to modulate the fluidity and stability of membranes, and reduces membrane permeability. Cholesterol interacts with the other constituents of mammalian cell membranes (e.g., lipids and proteins), and therefore water-soluble molecules that bind cholesterol need to compete with these membrane components (Figure 1). When the concentration of cholesterol increases and the interactions with other membrane components are saturated, cholesterol becomes more accessible to water-soluble molecules. Cholesterol accessibility varies greatly with the phospholipid composition of the membrane.
Cholesterol accessibility (and not overall cholesterol content) has been shown to be important for many physiologically processes that involve cholesterol binding and for cell signaling. Problems in cholesterol homeostasis have been shown to play a role in Alzheimer’s disease, heart disease, and diabetes among others. To date, there are no probes able to determine changes in the accessibility cholesterol at the membrane surface. PFO is an excellent candidate for the creation of a probe to measure cholesterol accessibility. The binding isotherm of PFO to cholesterol-containing membranes presents a sharp and sigmoidal response to the increase in cholesterol concentration. No binding is detected at low cholesterol levels, but when interactions with other components saturate, cholesterol becomes accessible and PFO binds. We have shown that modifications to the PFO binding domain can alter the affinity of the toxin for cholesterol (i.e., requires more or less cholesterol for binding). Combining PFO mutants with different cholesterol affinity with several fluorophores allow the examination of cholesterol accessibility in different cell membranes. This work, in collaboration with Professor Bernardo Trigatti from McMaster University, was recently published in the journal Biochemistry (Johnson, B.B., et.al. “Modifications in Pergringolisin O domain 4 alter the cholesterol concentration threshold required for binding” Biochemistry 2012 51 (16) 3373-82).
Address Goals
Our primary goal with this research is in the area of research infrastructure – the generation of a new tool for the detection of cholesterol accessibility at the surface of cell membranes. Through this work, we will discover the molecular mechanisms of how water-soluble molecules interact with membrane cholesterol and how cholesterol accessibility regulates cell physiology, enabling new strategies for the treatment of disease.