Highlight
Using Neutron Scattering to Track Gas Molecules Over the (CH4)1-x(CO2)x ? 5.75H2O Clathrate Hydrate Solid Solution
Achievement/Results
Michelle Everett, a National Science Foundation-funded Integrative Graduate Education and Research Traineeship (IGERT) successfully completed her Ph.D. in Materials Science and Engineering from the University of Tennessee Knoxville in May 2013. Her Ph.D. work was exemplary of the STAIR IGERT because it concerned sustainable technology, specifically the reduction and capture of CO2. Dr. Everett’s research contributed to a better understanding of how these materials can decompose, liberating methane when taken out of their pressure/temperature stability zone, and how the physical properties change when exchanging CH4 with CO2.
Gas hydrates have generated interest over a wide range of academic disciplines (Chemical Engineering, Biology, Earth and Planetary Sciences, etc.) and industry (mainly the gas and oil companies) due to not only the possibility that they could provide a large source of methane and could be a medium for sequestering carbon dioxide, but also for their role in the global carbon cycle and climate change as well as plugging gas pipelines, transporting methane, and their possible existence on other planets including Mars. The research team here was no exception and collaborators included researchers specializing in microbiology, geosciences, chemistry, physics, chemical engineering, and materials science and engineering. Gas hydrates are very challenging to work with since most of them are only stable at low temperature/high pressure conditions and need to kept at temperatures below 140 K or elevated pressures.
Dr. Everett used high resolution neutron powder diffraction to study the exchange of CO2 for CH4 in natural gas hydrates. The engineering goal includes both the aspects of harvesting methane from natural hydrate deposits while simultaneously sequestering industrially produced CO2. The process would involve pumping CO2 deep into the sediment layers where natural hydrates rich in CH4 are found and having the CO2 replace the CH4 in the hydrate structure, resulting in a release of CH4. However, for this theoretically simple process many engineering challenges exist requiring a detailed understanding of how the physical properties respond while exchanging the CH4 and CO2 molecules in the structure. This solid solution provides a platform to study how two very different molecules, C is linearly coordinated to the two terminating O atoms in the CO2 molecule while in CH4 the C is tetrahedrally coordinated to the four terminating H atoms can changed the properties of the framework where they are occluded. After successfully synthesizing gas hydrate samples with minimal additional ice using a technique of introducing a mixture of CO2/CH4 gas in an sealed pressure vessel containing water then subsequently cooling and tumbling the vessel, Everett applied for and was awarded beamtime on the POWGEN time-of-flight diffractometer at the Spallation Neutron Source at Oak Ridge National Laboratory.
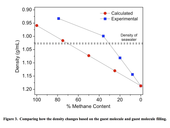
Everett is shown loading one of her samples into the POWGEN sample chamber in Figure 1. The high resolution powder diffraction data and advanced data analysis techniques used by Everett allowed her to construct neutron difference maps, allowing for the visualization of the gas molecules over the series. Additionally, Everett was able to quantify how the unit cell volume decreases as the CH4 is exchanged for CO2. Any volume change (either increasing or deceasing) can have implications for structures built near the reservoir where the exchange is taking place. Everett was also able to calculate the density from the refined lattice parameters and compositions of the samples studied, shown in Figure 3, to determine at what composition the gas hydrate would go, floating in sea water to sinking (CH4 gas hydrate floats while CO2 gas hydrate sinks). Additionally, Everett’s work on gas hydrates has resulted high quality atomic details that provide input for computational scientists and modelers working to better understand these materials that only exist at subambient temperatures and high pressures requiring special conditions to study them in the laboratory.
Address Goals
The research of Michelle Everett advances the frontiers of knowledge and provides a fundamental mechanism for understanding how methane can be replaced with carbon dioxide in gas hydrates in the Earth or at the bottom of the oceans. This process would simultaneously provide natural gas as well sequester carbon dioxide, a green house gas, resulting in a nearly carbon-neutral fuel supply.