Highlight
IDENTIFYING THE GEOMETRIC TRIGGERS FOR CELL STEMNESS
Achievement/Results
NSF funded IGERT trainee at Rutgers, Aaron Carlson, has advanced a new study on the role of substrate geometry on cell colony formation, self-renewal, and differentiation of pluripotent stem cells by using completely synthetic, three-dimensional fibrous microenvironments. This work recently appeared in the FASEB Journal (2012).
Human pluripotent stem cells (hPSCs), which include embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs), have incredible potential as an unlimited source of human cells for regenerative medicine, in vitro models of human disease, or in vitro drug screening applications. A key limitation hPSC culture is that most culture systems restrict hPSCs to two-dimensional (2-D) systems, which have a limited ability to mimic the complex three-dimensional (3-D) microenvironment inhabited by these cells during embryonic development. Thus, our ability to exert precise control over hPSC expansion, self-renewal, and differentiation remains limited.
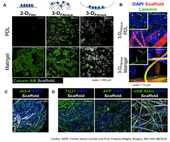
A major new IGERT research investigation was initiated by IGERT trainee Aaron Carlson in the laboratory of Professor Prabhas Moghe and PI, NSF IGERT on Stem Cell Science and Engineering, through extensive collaboration between several laboratories at Rutgers. Using the IGERT best practice of an IGERT advisory constellation of faculty experts, Carlson put together a team of co-advisory laboratories (Professors Joachim Kohn, Martin Grumet, Ronald Hart, Jennifer Moore, Rick Cohen). In addition, this project builds on the legacy of the work, training, and advice provided by several current and former IGERT students, including Charles Florek, Rebecca Moore, Jocie Cherry, Christopher Ricupero, and others. Aaron Carlson examined the critical determinants of hESC self-renewal in fully synthetic polymer-based substrates with variable microgeometries, including planar 2-D films, macroporous 3-D sponges, and microfibrous 3-D fiber mats. Polymer substrates were fabricated from poly(DTE carbonate) (pDTEc), a member of a combinatorial library of tyrosine-derived polycarbonates developed by the New Jersey Center for Biomaterials. He discovered that a minimal pretreatment with poly-D-lysine (PDL), a synthetic polycation, was sufficient for hESC adhesion, survival, and colony formation within fibrous scaffold configurations but not 2-D planar substrates or 3-D macroporous scaffolds. This is contrary to the vast majority of 2-D substrates that support hESC culture, which require either co-culture with fibroblast feeder layers or pretreatment with Matrigel. Interestingly, he found similar levels of hESC colony formation within PDL-treated fibrous scaffolds fabricated from different polymer chemistries, suggesting that substrate geometry rather than the specific pretreatment of polymer composition is a key factor dictating hESC survival. Through this process, Carlson identified a defined and exogenous extracellular matrix (ECM)-free substrate consisting of PDL-treated electrospun fibrous scaffolds that can support hESC survival similar to control cultures consisting of Matrigel-treated fibrous scaffolds. The central hypothesis was that a critical 3-D organization of hESC clusters within the fibrous scaffolds allows endogenous ECM and enables survival of these cells in the absence of exogenously added factors. Within fibrous scaffolds with large (~3.5 µm) fibers, hESCs infiltrated within the scaffold mesh, establishing extensive cell-cell and cell-matrix contacts within 7 days of seeding. Indeed, cells were found to not only produce copious amounts of ECM proteins laminin, collagen type IV, and collagen type I in PDL-treated scaffolds, and but also deposit these proteins within cell clusters. This finding was in stark contrast to hESCs in traditional Matrigel-treated scaffolds, for which endogenous ECM appears absent due to exclusive localization of ECM ligands to the scaffold fibers (where they are adsorbed prior to cell seeding). This confirms our hypothesis that matrix-production occurs within PDL-treated fibrous scaffolds.
We next investigated whether binding to one key protein, endogenous laminin, was important for initial hESC adhesion and survival. Functional inhibition of laminin in fibrous scaffolds treated with PDL or Matrigel and 2-D controls treated with Matrigel all demonstrated decreased numbers of viable cells relative to Isotype controls. While this is expected in Matrigel-treated substrates (which contain high levels of laminin), inhibition of hESC survival in PDL-treated scaffolds suggests that binding to endogenously produced laminin is critical to hESC survival. Carlson further investigated the range of hESC behaviors that could be demonstrated within these exogenous matrix-free substrates. He assessed hESC proliferation and self-renewal after 7 and 14 days within our scaffolds and found that two different hESC lines displayed similar proliferation and similar levels of pluripotency markers and pluripotency genes to 2-D Matrigel controls. Finally, he demonstrated directed differentiation in situ to neuronal cells, hepatic lineage cells, and smooth muscle cells similar to Matrigel-treated controls. This demonstrates that endogenous ECM is sufficient for hESC self-renewal and directed differentiation within synthetic 3-D microenvironments.
Address Goals
There are several potential applications and implications for this work related to both the DISCOVERY goals and RESEARCH INFRASTRUCTURE goals. First and foremost, this work demonstrates that 3-D microenvironments can obviate the need for complex ECM pretreatments traditionally required for hESC culture and differentiation. Eliminating such ECM from hESC substrates is an important step towards improving the reproducibility and clinical relevance of hESC culture systems. This system thus may further be developed into a synthetic substrate for hESC expansion/propagation. In addition, our differentiation studies suggested that the matrix composition of the substrate can influence differentiation to different lineages. Further optimization could yield improved protocols for efficient generation of differentiated cells for various applications. This system is particularly amenable to regenerative medicine, since scaffolds containing differentiated cells can potentially be direct transplanted in vivo. In addition, as the Rutgers team has recently demonstrated that iPSCs behave similarly to hESCs within our PDL treated fibrous scaffolds, this system may be adapted as a tool for in vitro disease modeling via patient-specific iPSCs or in vitro drug screening or toxicity testing for a variety of applications in a more “in vivo-like” 3-D microenvironment.