Highlight
Collaborative efforts support critical work for understanding material/biological interactions
Achievement/Results
Over the past year, University of Oregon doctoral student Ian Moody used the resources of his National Science Foundation IGERT Traineeship to increase participation in a collaboration with Oregon State University Professor Robert Tanguay’s Biology/Toxicology research group. Moody, who studies materials chemistry in the lab of UO Professor Mark Lonergan, wished to use this opportunity to expand the scope of his research to include not just design of new materials, but also understanding the ramifications for utilizing such materials.
One research thrust in the Tanguay lab is to evaluate biological interactions and responses to nanoparticles. Researchers then seek to understand the mechanisms by which exposures produce biological responses as this puts them in a strong position to develop strategies to protect both humans and the environment. A challenge in understanding the toxicology of nanomaterials is correlating observed biological effects to specfic physico-chemical features of the material. Toxicity can just as often be the result of impurities or decomposition products as from the actual material under study. Toxicology labs often do not have the resources or expertise to fully characterize their materials, and thus rely on the physical data provided to them by the manufacturer. Interdisciplinary collaborations such as this one between the Lonergan Lab and the Tanguay Lab – are, therefore, crucial for understanding these complex material/biological interactions.
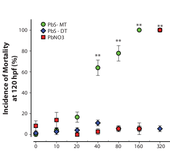
In this collaboration, a series of toxicology experiments were performed on embryonic zebrafish as they were exposed to lead sulfide nanoparticles (PbS-NPs). PbS-NPs have unique optical and electrical properties – in particular, sensitivity to near-infrared (NIR) light – that have made them compelling candidates for a variety of applications, including NIR-light-emitting diodes (LEDs) and photovoltaic cells. The goal of this study was to measure the toxicity of PbS-NPs as a function of ligand shell composition, and more specifically, highlighting the importance of the ligand head group. Additionally, these types of studies aim to identify design rules that apply more generally to nanomaterials, with the goal of producing robust nanoparticle systems that are less toxic and more environmentally benign.
Results from multiple exposure trials indicate that subtle changes in ligand shell composition can have large influences on biological interactions. Specifically, PbS-NPs that were functionalized with 3-mercaptopropanesulfonate (PbS-MT) ligands elicited higher levels of mortality in embryonic zebrafish than PbS-NPs functionalized with 2,3-dimercaptopropanesulfonate (PbS-DT) ligands (see Figure). This difference in biological response is surprising because the two ligands are nearly identical in structure, with the only structual difference being the ligand head group – a monothiol for MT and a dithiol for DT. In a previous paper (Moody 2008), Moody showed that PbS-MT nanoparticlesare less stable to oxidative decomposition than PbS-DT nanoparticles, which suggests a connection between physical degradation of the nanoparticles and the toxic response. Since degradation is believed to occur in tandem with ligand desorption, it was important to confirm that toxicity was not simply due to the ligand oxidation product. Controls run with ligand-only solutions showed benign behavior, confirming that toxicity is tied to the nanoparticle-ligand complex.
One of the challenges of this study has been understanding the physical process by which PbS-MT and PbS-DT nanoparticles trigger the toxic response in zebrafish. Inductively-coupled plasma mass spectrometry (ICP-MS) measurements on exposed embryos show that some form of lead is able to enter the fish, and cellular death studies confirm that tissue damage occurs upon exposure. To test whether the ligands are simply sequestering lead-containing, decomposition products, a series of toxicity exposures were performed with a soluble lead control sample (lead nitrate) and varying amounts of ligands. Greater ligand-to-Pb ratios result in reduced toxicity for both ligands, and DT reduces toxicity more significantly than MT.
Finally, the soluble lead content of solutions of PbS-MT and PbS-DT nanoparticles were monitored over time via ICP to determine if there was a difference in the protective qualities of the ligands. PbS-MT nanoparticles were found to release a greater amount of lead into solution than PbS-DT.
The picture that emerges from this data is one in which the DT ligand interacts more strongly with the PbS-NPs than athe MT ligand, protecting them from decomposition, and making them less bioavailable to the zebrafish. This study is now in its final phases, with final edits being made on a manuscript that will be submitted for publication.*
*Truong, L., Moody, I.S., Lonergan, M.C., Tanguay, R.L. (2010). “The Ligand Head Group of Lead Sulfide Nanoparticles Influences Biological Responses in Embryonic Zebrafish.” Arch Toxicol (manuscript in preparation)
Address Goals
This research activity advances knowledge into an area of toxicology with respect to new materials. This research is critical as we seek to advance fundamental knowledge of science and protect our environment.
The cross-university, cross-specialty collaborative nature of this activity makes it the sort of out-of-the box training activity exemplified by the IGERT program.