Highlight
Targeting of mRNAs with branched peptides
Achievement/Results
MILES faculty Dr. Webster Santos and MILES trainee David Bryson are investigating peptides that have potential health properties. After screening out 16 HIV-1 TAR RNA binding “hit”-compounds from a branched peptide library comprised of 4,096 members and generating an additional optimized sequence, the compounds were synthesized and purified for validation. The prepared compounds were reverse phase HPLC purified and confirmed by MALDI-TOF analysis. These were sent to the lab of Dr. David Rekosh at UVA for in vitro studies to determine their efficacy at inhibiting viral replication of the HIV-1 virus in a luciferase based model system. Branched peptide (BP) 15 = (RRA)2 KVRD was successful at significantly inhibiting viral replication of two different clades of HIV in their system. The effect was also shown to be dose dependent.
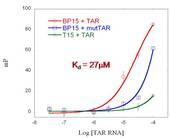
This is an exciting development and shows that our branched peptides may have promise as HIV-1 therapeutics in the future. A second batch of the 17 branched peptides were synthesized with one N-terminus capped by an acetyl group, and the second N-terminus capped by the fluorophore, fluorescein isothiocyanate (FITC). A linear variant of BP15 was also prepared (RRAGVRD = T15) and FITC labeled to serve a control in our experiments. This linear control was used in a fluorescence polarization assay (Figure 1) to compare to its binding kinetics with BP15 (Kd= 27 M). The reduced affinity of T15 for the HIV-1 TAR RNA compared to BP15 supported our initial hypothesis that by including a branching unit, a strong multivalent interaction with the target would be generated. The FITC labeled branched peptides were also used in cell permeability, flow cytometry and MTT assays by Patrick McLendon in the lab of Dr. Reineke at Virginia Tech. The branched peptides were determined to have low toxicity, and good cell permeability. T15 displayed little to no cell permeability.
Address Goals
Due to high cost and difficulty in conducting the fluorescence polarization (FP) assays, researchers have begun characterizing binding kinetics in all of the FITC labeled branched peptide by dot blot assays (Figure 2) using 32P labeled HIV-1 TAR RNA. These assays demonstrate significantly lower Kd values than the corresponding FP assays, where the new Kd values are in the middle to high nanomolar range. This is encouraging because the branched peptides have a strong affinity for the therapeutic target.