Highlight
Scorpion Venom With Nanoparticles Halt Brain Cancer Spread
Achievement/Results
A University of Washington (Seattle) interdisciplinary team of biologists and materials engineers, including both current and former IGERT trainees and led by UW Professor Miqin Zhang, have developed a nanoparticle aimed at slowing the spread of brain cancer. By combining nanoparticles with a scorpion venom compound already being investigated for treating brain cancer, their research has found that they could cut the spread of cancerous cells by 98 percent, which was almost 100 percent improvement of the scorpion venom compound treatment alone.
For more than a decade scientists have looked at using chlorotoxin, a small peptide isolated from scorpion venom, to target and treat cancer cells. Chlorotoxin binds to a cell surface protein complex MMP-2, which is overexpressed by many types of tumors, including brain cancer. Previous research by Zhang’s team combined chlorotoxin with special nanometer-scale particles, which can be visualized by both magnetic resonance (MR) and intra-operative optical imaging. This imaging contrast agent is being tested as a tool to aid neurosurgeons in surgical removal of brain tumors.
Chlorotoxin can also induce a therapeutic effect by disrupting the spread of invasive tumors – specifically, it slows cell invasion, the ability of the cancerous cell to penetrate the protective matrix surrounding the cell and infiltrate far in to healthy brain. The chlorotoxin molecule is believed bind cells by way of the MMP-2 protein complex, which is hyperactive in highly invasive tumors such as brain cancer. Researchers believe MMP-2 helps the cancerous cell break through the protective matrix to invade new regions of healthy brain. But when chlorotoxin binds to MMP-2, both get internalized into the cancerous cell.
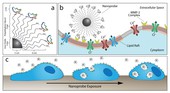
In their new study Zhang’s team investigated the therapeutic efficacy of their chlorotoxin-decorated nanoparticles and found the resultant complex doubles the therapy’s effect compared to chlorotoxin alone. The use of nanoparticles in combination with therapeutics has been shown to improve treatments, because the pairing lasts longer in the body, offering a better chance of reaching the tumor. Here Zhang’s team demonstrated that the combination also boosts the effect because 10 therapeutic chlorotoxin molecules are spatially arranged around each nanoparticle enabling numerous simultaneous interactions with MMP-2 protein complexes on cancer cell surfaces. An illustration of how these nanoparticles interact with MMP-2 protein complexes on surface of cancer cells and elicit their therapeutic effect is shown in figure 1.
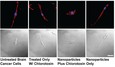
A series of imaging studies showed that the nanoparticles were rapidly internalized, consequently lowering the number of active MMP-2 groups at the cell surface and inhibiting cell motility. Shown in figure 2 is a transmission electron microscope (TEM) image of a chlorotoxin-decorated nanoparticle in the process of interacting with the surface of a brain cancer cell and subsequently being internalized.
Further imaging studies showed that the cells exposed to chlorotoxin-decorated nanoparticles were unable to elongate, whereas cells containing only nanoparticles or only chlorotoxin could stretch out (Figure 3). These results suggest that the chlorotoxin-decorated nanoparticles were able to disable the machinery on the cell’s surface that allows cells to change shape, yet another step required for a tumor cell to slip through the brain.
The findings of this research were published in the January 19, 2009 issue of the journal Small, and subsequently featured on over 1,000 websites and news outlets including the National Geographic News, United Press International, and Science Daily.
Along with Professor Miqin Zhang other co-authors of the published study are Omid Veiseh, Jonathan Gunn, Forrest Kievit, Conroy Sun and Chen Fang of the University of Washington (UW) and Jerry Lee of the National Cancer Institute and Johns Hopkins University. This research was funded by the National Institutes of Health, and through traineeships from the National Science Foundation (NSF) Integrative Graduate Education and Research Traineeship Program (IGERT)-Program PI Marjorie Olmstead UW.
Address Goals
So far most cancer research has combined nanoparticles either with chemotherapy that kills cancer cells, or therapy seeking to disrupt the genetic activity of a cancerous cell. This is the first time that nanoparticles have been combined with a therapy that uses the unique properties of nanomaterials to limit the spread of a cancer cell.
Slowing the spread of cancer with chlorotoxin nanoparticle complexes would be especially useful for treating highly invasive tumors such as brain cancer, but it’s application is not limited. MMP-2 may also be overactive in prostate, colon, skin, lung, and ovarian cancer, and Zhang’s team believe that the technique could slow the spread of these other tumors.
The team is currently seeking industry partnerships to bring this promising research to the clinic, and hopes that trials could begin within the next 8-10 years.
Students working on this project gain tremendous experience bridging disciplines from the materials and chemical engineering required to fabricate the nanoparticles through the bioengineering required to modify their biocompatibility to medical applications, currently in rodents, but looking forward to human medicine. All four students involved in this project are (or were) enrolled in the interdisciplinary Ph.D. program developed under this IGERT, and two have been directly supported by IGERT traineeships.