Highlight
Optically guided controlled release from liposomes with tunable plasmonic nanobubbles.
Achievement/Results
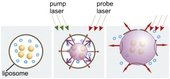
Accurate delivery of therapeutics is one of the biggest challenges of medicine in general. This has lead to the development of a number of approaches to encapsulate the therapeutic load in a delivery vehicle that can release the load at the disease site. The team from Rice University lead by Dr. Dimitri Lapotko and Prof. Jason Hafner has developed a new method to optically determine the accumulation of liposomes loaded with therapeutic agents and optically control the release of the molecular load from liposomes using plasmonic nanobubbles. Gold nanoparticles are extremely efficient in converting optical energy into thermal energy and act as the source of the plasmonic nanobubbles when irradiated with light close to the plasmon resonance of the gold nanoparticles. In the current series of experiments to demonstrate the technique, liposomes (<1 micron in size) were loaded with two different fluorescent protein molecules and 80 nm gold nanoparticles. These liposomes were then immobilized on a glass slide.
A pulsed laser at 532 nm for 0.5 ns was used to excite the nanoparticles. The thermal energy from the nanoparticles is transferred to the surrounding medium and a nanobubble is formed around the NPs. The size of the plasmonic nanobubble depends upon the fluence from the laser on the nanoparticles. Nanobubbles scatter light more efficiently than the gold nanoparticles and so scattering measurements can be used to determine the size of the bubble and the location of the liposome. A laser pulse can be used to generate a plasmonic nanobubble greater than the size of the liposome to disrupt the liposome membrane and release the molecular load. The released fluorescent proteins can be monitored by fluorescence spectroscopy.
The size of the nanobubble generated depends upon the laser fluence and the number of nanoparticles in the liposome. Aggregates of a few or more nanoparticles were demonstrated to form larger bubbles and at lower pump laser fluence. Next the lifetime of the nanobubbles for different size nanobubbles was determined. An almost linear relationship between the laser fluence, size of the nanobubbles and lifetime exists beyond a threshold fluence. The plasmonic nanobubble generation is reproducible when more than one pulse was applied to the same liposome. Therefore several sequential plasmonic nanobubbles of varying sizes can be generated on demand.
To study the release of the molecular load from the liposome, the fluorescence from the fluorescent protein inside the liposome is monitored along with scattering from the plasmonic nanobubbles. Fluorescence imaging before the pump pulse shows the fluorescence concentrated within the liposome. Fluorescence imaging after the pump pulse used to generate the disruptive nanobubbles shows a concentric halo outside the liposome, and the fluorescence inside the liposome drops. This is direct evidence of the rupture of the liposome and release of the proteins.
In this research achievement a new technique for molecular load delivery combining plasmonics with liposome delivery was developed. The plasmonic nanobubble release method has several unique features: the mechanism is mechanical, non thermal, fast (in milliseconds), local and tunable; the release agent the nanobubble is not a particle but a transient on demand event that combines mechanical and optical properties in one agent; uses biocompatible gold nanoparticles; and the release mechanism does not depend upon a biochemical or physiological factor that could trigger unintended release. The techniques developed as a result of the research are not unique to liposomes but maybe generalized to other carrier capsules.
The team at Rice University also developed an international collaboration with the A.V. Lykov Heat and Mass Transfer Institute, Belarus.
Address Goals
In our IGERT program we are expanding the field of nanophotonics towards new applications. This requires a multidisciplinary outlook to combine optics and plasmonic properties of nanoparticles to overcome limitations in biological systems. As demonstrated in the research achievement, by exploiting the plasmonic photothermal properties of gold nanoparticles new techniques in controlling the release of therapeutic loads in cells have been developed.
The secondary NSF goal of training the next generation of scientists and engineers with the multidisciplinary outlook is achieved in training graduate and undergraduate students. Lindsey Anderson the lead author in the research is a graduate IGERT trainee. Dr. Lapotko trained Lindsey to use the photothermal microscope to study this process, and to demonstrate the disruptive effects of the nanobubbles. According to Lindsey the hardest part of the experiment was separating the data from artifacts due to scattering from dust particles. She worked with Kate Hleb at the A.V. Lykov Heat and Mass Transfer Institute for analyzing the nanobubble data. She worked with Steve Hansen a high school student who spent the summer working alongside her.